Production-grade deliverables with executive summaries, multi-endpoint scoring tables, ICH alignment documentation, and three-layer forensic verification. Every claim evidence-backed.
Two representative reports from recent client engagements. Actual report structure and depth, with client-specific data redacted.
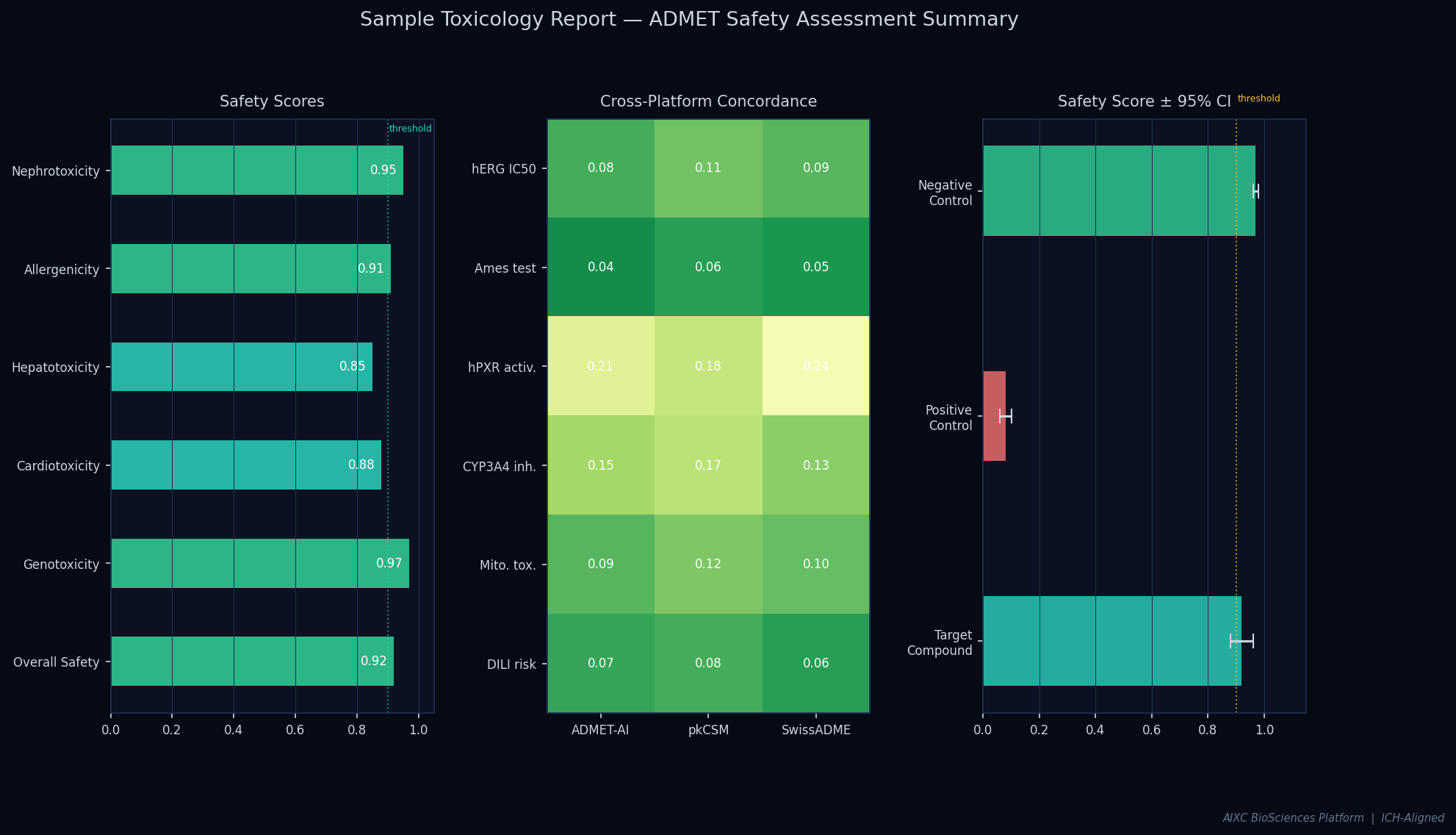
Comprehensive toxicology assessment for a novel small-molecule therapeutic candidate, culminating in an Ethical Committee submission that documents genomics and allergenomics coverage per OECD standards.
| Endpoint | Verdict |
|---|---|
| hERG Inhibition | PASS |
| Hepatotoxicity | PASS |
| Mutagenicity (Ames) | PASS |
| CYP450 Panel | PASS |
| Mitochondrial Toxicity | PASS |
Nonclinical overview for regulatory submission. ICH S9 aligned dose justification, safety pharmacology assessment, reproductive toxicology summary.
| Section | Status | Pages |
|---|---|---|
| Dose Justification | COMPLETE | 12 |
| Safety Pharmacology | COMPLETE | 8 |
| Genotoxicity Assessment | COMPLETE | 6 |
| Reproductive Toxicology | COMPLETE | 10 |
| ICH S9 Alignment | VERIFIED | 4 |
| Cross-Reference Matrix | COMPLETE | 3 |

Clear, actionable overview for decision-makers. Key findings, risk assessment, and recommendations in plain language.
Multi-endpoint quantitative scores with confidence intervals, model ensemble agreement, and pass/fail classifications.
Explicit mapping to ICH guidelines (M7, S1A/B, S2, S9). Regulatory-ready documentation for IND/IMPD submissions.
Three-layer verification: internal consistency, cross-platform concordance (r>0.99), and literature validation.
Every claim passes internal consistency checks, cross-platform concordance validation, and primary literature verification.
Results validated across multiple independent computational platforms. Pearson correlation exceeding 0.99 across all endpoints.
All reports aligned with current ICH guidelines. Ready for inclusion in IND, IMPD, and BLA regulatory submissions.
Standard toxicology screens delivered within 24 hours. Full pipeline reports within 2-4 weeks depending on scope.
Enter your details to receive a complete sample toxicology report demonstrating our methodology, depth, and presentation quality.
Tell us about your compound, target, or therapeutic program. We will scope a deliverable tailored to your needs.